- Dror Lindner, M.D.a,
- Kinzie G. Sharp, M.S., P.A.-C.a,
- Anthony P. Trenga, B.A.a,
- Jennifer Stone, M.A.a,
- Christine E. Stake, M.A.a,
- Benjamin G. Domb, M.D.a, b,
 ,
, 
- a The American Hip Institute, Chicago, U.S.A
- b Hinsdale Orthopaedics (K.G.S., C.E.S., B.G.D), Hinsdale, Illinois, U.S.A
- http://dx.doi.org/10.1016/j.eats.2012.10.002, How to Cite or Link Using DOI
- Permissions & Reprints
Abstract
The ligamentum teres (LT) has been studied since the 19th century, and its anatomy and biomechanical function have been well described. Recent advancements in hip arthroscopy have caused increased awareness of LT pathology. Previous reports have estimated the incidence of LT tears during hip arthroscopy to be 4% to 51%, and LT tears have been estimated to be the third most common reason for hip pain in athletes. Biomechanical studies have shown the LT's role in stability of the hip. Despite the growing body of literature on LT anatomy and function, its role as a causative factor in hip pain and hip instability has yet to be clearly defined, and the treatment of LT tears remains controversial. However, in certain cases where hip subluxation and overt instability are related to a traumatic full-thickness tear of the LT, reconstruction of the ligamentum has been suggested. We describe a technique for arthroscopic LT reconstruction using either a semitendinosus autograft or allograft in the supine position.
The ligamentum teres (LT) is pyramidal in shape and arises from the transverse acetabular ligament and the posteroinferior portion of the acetabular fossa. Since the 19th century, its anatomy and mechanical properties have been studied, and the LT has been found to be as strong as the anterior cruciate ligament.1 Histologic studies have found that it possesses both mechanoreceptors and nociceptors,2 and 3 suggesting that the LT plays a part in the integral reflex system that is involved in joint protection, sense of pain, and proprioception.
In past reports, LT tears have been described after hip dislocations and traumatic twisting injuries to the hip. The prevalence of LT tears in hip arthroscopy was reported to be 5% to 17.5% in patients undergoing hip arthroscopy; in contrast, a recent article by Botser et al.4 documented LT tears in 51% of patients undergoing hip arthroscopy. However, this difference in prevalence may be attributed to an increased awareness and inclusion of low-grade partial tears in the study.
How the LT contributes to normal hip biomechanics and stability is still debatable. Some surgeons view the LT as having a negligible role in hip biomechanics and stability; for example, open surgical dislocations require transection of the ligament and are therefore based on the premise that sacrifice of the LT is inconsequential. In contrast, others believe that its role in normal hip biomechanics and stability warrants its reconstruction. Importantly, reconstruction has been described by Simpson et al.,5 who noted that LT reconstruction is in its infancy and requires further study and outcomes data. We have found that in a very specific group of patients, traumatic LT tears are associated with hip pain and instability. In such cases, where deficiency of the LT has altered the biomechanics of the joint, we believe that simple debridement will not alleviate all the symptoms and the patient will keep having pain and symptoms of hip instability. In these patients, reconstruction of the LT is warranted.
In development of this technique, numerous procedures were performed in cadaveric hips and were followed by open dissections to assess the proximity of the bone tunnels to the obturator vessels and identify additional challenges. The previously described technique was used as a basis.5 The result was a technique that was found to be both feasible and reproducible. We describe our technique for reconstruction of the LT in this report. Table 1 describes the pearls and pitfalls of this technique.
Table 1. Pearls and Pitfalls of Arthroscopic LT Reconstruction
|
Pearls |
Pitfalls |
| Careful patient selection is warranted. | Ensure that the graft is prepared before acetabular drilling, to provide quicker placement to minimize fluid entry into the pelvis and the chance for abdominal compartment syndrome. |
| Cadaveric models should be used before surgery. | Avoid penetrating the medial acetabular cortex with the guidewire during guidewire placement in the fossa. |
| Femoral tunnel: A free-hand technique is used under fluoroscopy or with an aimer device. | Avoid plunging into the pelvis during acetabular rimming. |
| Acetabular tunnel position:Internal rotation and abduction are required for correct tunnel placement. The tunnel must be in the most inferior part of the acetabular fossa but angled away from the obturator vessels. | During acetabular fixation with the RetroButton, avoid flipping the button too early and ensure that the button is secure to avoid loosening. |
| Acetabular tunnel drilling: The guidewire should be advanced to the second cortex without penetrating it; care is used while reaming. | Avoid over-tightening the graft. |
| Upon drilling the 2 tunnels, the graft should be passed as soon as possible to prevent fluid accumulation in the pelvis. | |
| Acetabular graft fixation: Passing and flipping the cortical button are challenging. Once the button has been flipped and secured, fluoroscopy should be used for verification. | |
| Rehabilitation: The patient is placed in a hip brace. Physical therapy is started on the first postoperative day, refraining from abduction and external rotation the first 6 wk. |
Technique
Patient Positioning and Portals
Under general anesthesia, the patient is placed in the supine position on a traction table or fracture table, both feet are well secured and padded, and a peroneal post is used to protect the genitalia. The hip is prepared and draped in the usual fashion. Traction is applied; a spinal needle, under fluoroscopic visualization, is inserted into the joint; and the joint is vented. After venting of the joint, additional traction is applied. The needle is retracted, and .5% bupivacaine is injected into the subcutaneous tissues. Correct portal placement is located by fluoroscopic visualization. The anterolateral portal is first established by use of a No. 11 blade to make an incision in the skin. A spinal needle is inserted through the incision and into the joint, with care taken to avoid the labrum and the femoral head. An over-the-guidewire technique is used to insert a 70° arthroscope through a 4.5mm cannula. A mid-anterior portal is established by use of the same over-the-guidewire technique. The surgeon uses a beaver blade to perform a capsulotomy, incising the capsule parallel to the acetabular rim to connect the 2 portals.
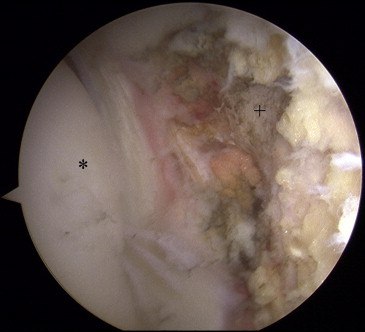
A diagnostic arthroscopy is performed, switching portals frequently to access all parts of the joint. Any additional pathology in the joint is addressed before LT reconstruction. The LT is examined and probed upon identification of a complete tear (Fig 1 and Video 1); the stump in the acetabular fossa is cleared with the Nav X ablation device (Arthrex, Naples, FL) and a shaver (Fig 2).
Fig 1. Arthroscopic view of left hip showing complete tear of LT. The patient is in the supine position, with visualization of the central compartment of the hip through the anterolateral portal. (Asterisk, femoral head; plus sign, LT stump.)
Fig 2. Arthroscopic view of left hip after debridement of LT stump from fossa. The patient is in the supine position, with visualization of the central compartment of the hip through the anterolateral portal. (Asterisk, femoral head; plus sign, fossa.)
Graft Preparation
The graft choice may include a semitendinosus autograft or allograft. Our preference is to use autograft. The double-stranded graft is prepared with maximal length on a RetroButton (Arthrex) with the graft sutured to the button by use of a 3-mm loop. The graft should be prepared before tunnel preparation.
Femoral and Acetabular Tunnels
A lateral 2-cm incision is made to approach the femoral transtrochanteric tunnel; the location is determined by fluoroscopy. A 3.2-mm guidewire (Arthrex) is passed through the lateral cortex of the greater trochanter, exiting through the center of the fovea in the footprint of the LT. This is performed in a “free-hand” technique with fluoroscopic assistance and direct visualization of the guide's exit point in the fovea. Over the guidewire, a cannulated reamer (Arthrex) is used to create the femoral tunnel. The reamer size used is determined by the graft size, which is measured during graft preparation. A shaver and wand are inserted through the femoral tunnel to complete the preparation of the footprint at the base of the acetabular fossa.
Drilling of the acetabular tunnel is performed through the femoral tunnel, with the hip internally rotated and abducted to achieve correct tunnel positioning in the cotyloid fossa. The anatomic insertion of the LT in the cotyloid fossa is made in the inferior portion of the fossa. To maintain a safe distance from the obturator vessels, the tunnel is placed slightly posterior to the center of the base of the fossa. The guidelines for safe acetabular drilling are well established and have long been in use by surgeons performing hip arthroplasty for screw placement in the acetabular component. The safe zones were first described by Wasielewski et al.,6 and 7 who suggested that the posteroinferior and posterosuperior quadrants are safe for drilling whereas the anterior quadrants are not. The structures at risk while drilling in the posteroinferior portion of the cotyloid fossa are the obturator artery and vein; however, they are at a safe distance from the exit point of the drill.5
A guidewire (Arthrex) is placed through the femoral tunnel and into the posteroinferior portion of the fossa; its position is verified by direct visualization. Once the correct position has been achieved, the guide is drilled to the medial cortex, without penetrating it, with fluoroscopic assistance (Fig 3). Over the guidewire, a cannulated reamer (Arthrex) is used to create the acetabular tunnel. The reamer used is determined by the graft size, which is measured during graft preparation. Fluoroscopic assistance is used to ensure that the guidewire is not penetrating into the pelvis, and the drilling is performed cautiously to avoid plunging through the medial cortex of the acetabular fossa.
Fig 3. Fluoroscopic intraoperative image, anteroposterior of hip, with hip in internal rotation. After reaming of the femoral tunnel over a guidewire, the guidewire is passed through the femoral tunnel into the desired location of the acetabular tunnel. The hip is placed in internal rotation to accommodate correct placement of the guidewire. The guidewire is drilled into the acetabular footprint, with care taken not to penetrate the medial cortex of the acetabulum.
Graft Placement
Once both tunnels are prepared, the graft is passed through the tunnels. Graft placement is performed by direct visualization and fluoroscopic assistance. Two knot pushers are used to lead the graft-button complex through the tunnels; one knot pusher is used to lead the button through the tunnel, and the second knot pusher is used to flip the button over the medial cortex. Once the button has been flipped, tension is placed on the graft and fluoroscopy is used to ensure that the button has flipped and is secure.
The motion and tension of the graft are examined in internal and external rotation while the hip is in traction. The traction of the leg is then removed while traction is maintained on the graft. The leg is positioned in 10° of hyperextension and 60° of external rotation, and a polyetheretherketone interference screw (Arthrex) is used for femoral fixation. Once the graft is secured, traction is reapplied, the arthroscope is reintroduced, and the graft is again examined (Fig 4) throughout the range of motion.
Fig 4. Arthroscopic view of central compartment of left hip through anterolateral portal after graft fixation, evaluating graft through range of motion. (A) Hip in neutral position. (B) Hip in external rotation. (Asterisk, femoral head; plus sign, LT.)
The excess graft is cut flush with the lateral cortex of the femur. The portals are closed with No. 3-0 Monocryl (Ethicon, Somerville, NJ), and the distal incision is closed with No. 1 Vicryl (Ethicon) for the fascia, No. 2-0 Vicryl for the subcutaneous layer, and No. 3-0 Monocryl for the skin. The patient undergoes placement of an X-Act ROM hip brace (Fig 5) (DJO Global, Vista, CA) and abduction pillow.
Fig 5. Fitting a right hip brace.
Rehabilitation and Recovery
For the first 6 weeks, the patient is kept in a hip brace (DJO Global) locked at 0° to 90° of flexion at all times and is restricted to 20 lb of foot-flat weight bearing. In addition, an abduction pillow is used at night for the same period. The patient starts physical therapy on the first postoperative day and is instructed to refrain from adduction and external rotation. Six weeks postoperatively, use of the brace and crutches is discontinued and the patient continues physical therapy with an emphasis on strengthening the gluteus medius and core muscles, as well as gradual progression of range of motion.
Discussion
The LT has been studied since the 19th century; however, only in recent years have we started understanding its true function. Today, we know that the LT is as strong as the anterior cruciate ligament; is tight in flexion, external rotation, and abduction; and plays a role in hip proprioception and pain (nociceptors and mechanoreceptors).1
The current standard surgical treatment for LT tears consists of debridement. Byrd and Jones8 reported on 23 patients who underwent debridement of LT tears with good outcomes. However, 15 patients in their study group had additional pathologies that were addressed during surgery. Haviv and O'Donnell,9 in a series of 29 patients, reported on isolated LT tears, reporting improvement of 16 points in the modified Harris Hip Score; however, 5 patients (17%) required revision surgery. Despite the good results reported with debridement, there appears, from the previous literature, to be a subset of patients who have residual pain and instability after LT debridement.
Currently, the indications for LT reconstruction have yet to be clearly established; however, it may be considered for patients with isolated complete LT tears who report subjective hip instability and have increased external rotation.
We have found this technique of LT reconstruction to be safe and reproducible. Further studies on LT reconstruction need to be conducted to establish the exact role and recommendations for it in the treatment of LT tears.
Supplementary Data
Video 1. ▪▪▪
Help with MOV files
Options
References
- D. Wenger, F. Miyanji, A. Mahar, R. Oka
- The mechanical properties of the ligamentum teres: A pilot study to assess its potential for improving stability in children's hip surgery
- J Pediatr Orthop, 27 (2007), pp. 408-410
- [SD-008]
- S. Sarban, F. Baba, Y. Kocabey, M. Cengiz, U.E. Isikan
- Free nerve endings and morphological features of the ligamentum capitis femoris in developmental dysplasia of the hip
- J Pediatr Orthop B, 16 (2007), pp. 351-356
- [SD-008]
- M. Leunig, M. Beck, E. Stauffer, R. Hertel, R. Ganz
- Free nerve endings in the ligamentum capitis femoris
- Acta Orthop Scand, 71 (2000), pp. 452-454
- [SD-008]
- I.B. Botser, D.E. Martin, C.E. Stout, B.G. Domb
- Tears of the ligamentum teres
- Am J Sports Med, 39 (2011), pp. 117S-125S
- [SD-008]
- J.M. Simpson, R.E. Field, R.N. Villar
- Arthroscopic reconstruction of the ligamentum teres
- Arthroscopy (2011), pp. 436-441
- [SD-008]
- R.C. Wasielewski, L.A. Cooperstein, M.P. Kruger, H.E. Rubash
- Acetabular anatomy and the transacetabular fixation of screws in total hip arthroplasty
- J Bone Joint Surg Am, 72 (1990), pp. 501-508
- [SD-008]
- R.C. Wasielewski, D.D. Galat, K.C. Sheridan, H.E. Rubash
- Acetabular anatomy and transacetabular screw fixation at the high hip center
- Clin Orthop Relat Res, 438 (2005), pp. 171-176
- [SD-008]
- J.W. Byrd, K.S. Jones
- Traumatic rupture of the ligamentum teres as a source of hip pain
- Arthroscopy, 20 (2004), pp. 385-391
- [SD-008]
- B. Haviv, J. O'Donnell
- Arthroscopic debridement of the isolated ligamentum teres rupture
- Knee Surg Sports Traumatol Arthrosc, 19 (2011), pp.1510-1513
- [SD-008]
The authors report the following potential conflict of interest or source of funding in relation to this article: American Hip Institute, Adventist Hinsdale Hospital, MedWest, and Arthrex.
 Address correspondence to Benjamin G. Domb, M.D., Hinsdale Orthopaedics, 1010 Executive Ct, Ste 250, Westmont, IL 60559.
Address correspondence to Benjamin G. Domb, M.D., Hinsdale Orthopaedics, 1010 Executive Ct, Ste 250, Westmont, IL 60559.